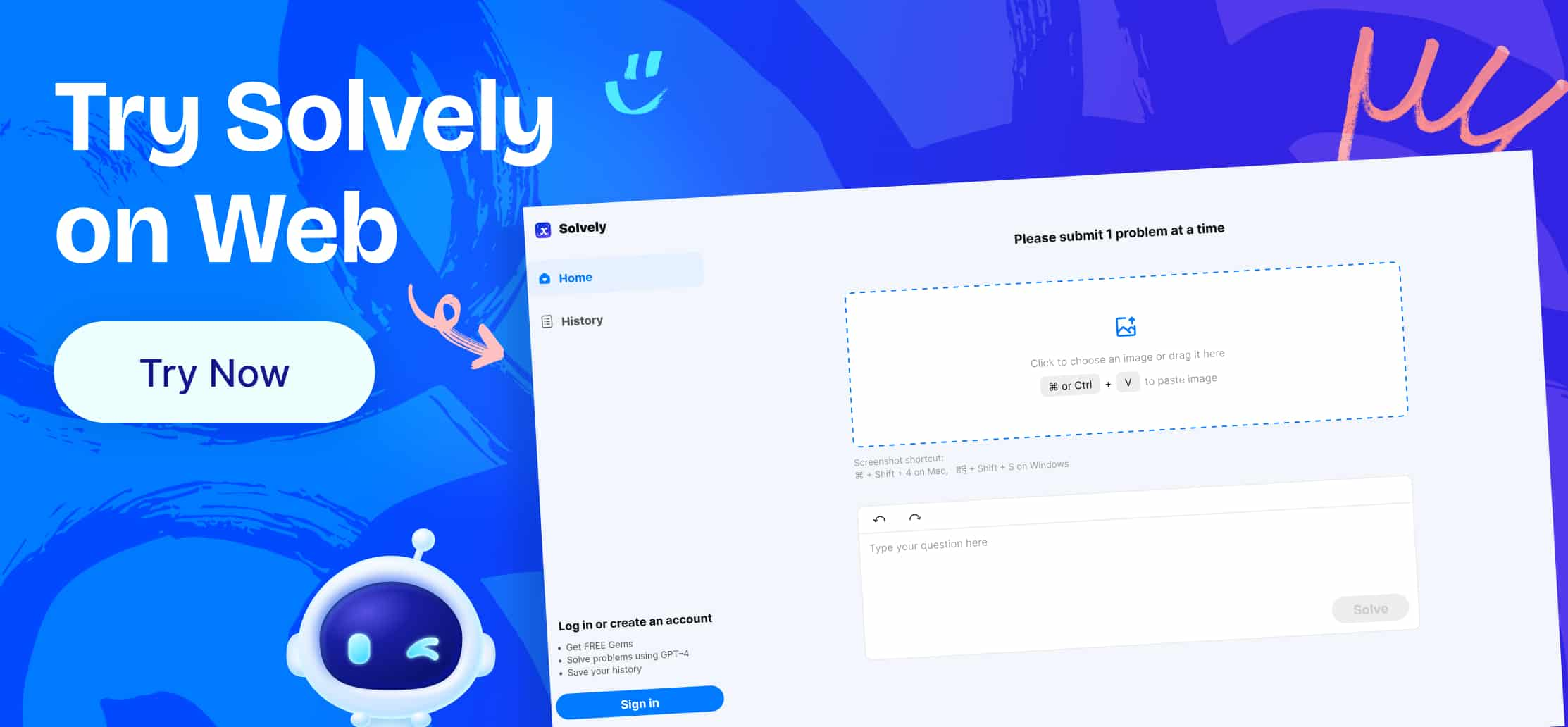
Tellurium ( $Z=52$ ) has a lower atomic number than iodine $(Z=53)$, yet it has a higher atomic weight (127.60 amu for Te versus $126.90 \mathrm{amu}$ for I).
Part A
How is this possible?
Tellurium has a greater atomic weight because its nuclei contain, on the average, more protons than an iodine nucleus.
Tellurium has a greater atomic weight because its nuclei contain, on the average, more neutrons than an iodine nucleus.
Tellurium has a greater atomic weight because its nuclei contain, on the average, more electrons and protons than an iodine nucleus.
Tellurium has a greater atomic weight because its nuclei contain, on the average, more electrons than an iodine nucleus.
Answer
\( \boxed{\text{Tellurium has a greater atomic weight because its nuclei contain, on the average, more neutrons than an iodine nucleus.}} \)
Step 1 :Since Tellurium has a lower atomic number than Iodine, it has fewer protons.
Step 2 :The difference in atomic weight must be due to the number of neutrons.
Step 3 :\( \boxed{\text{Tellurium has a greater atomic weight because its nuclei contain, on the average, more neutrons than an iodine nucleus.}} \)